RNA polymerase: the SARS-COV-2 “sloppy photocopier”

Publication date: 14-08-2021
Updated on: 16-05-2025
Topic: Covid-19
Estimated reading time: 1 min
Article Author
Eufemia PutortiMedical Editor
Massimo Degano
Editor and Translator
Viktoryia LuhakovaThe ongoing COVID-19 pandemic has forced the scientific community to devote itself to the research and development of various strategies to fight SARS-CoV-2 infection. The only informed way to identify new antivirals is through the knowledge of the three-dimensional structure, literally the shape, of the virus proteins that are responsible for cell infection and virus replication. Once the structure of proteins is known, it is possible for chemists to synthesize compounds that block its function, and consequently the virus life cycle.
We talk about it in this cycle of 5 episodes with the help of Dr. Massimo Degano, Group leader of the Biocrystallography Unit of the IRCCS San Raffaele Hospital and lecturer at our University (he teaches Chemistry at the Degree Course in Dentistry and Dental Implantology and at the International MD Program, Biochemistry at the Degree Course in Medicine, and Structural Biology at the Degree Course in Biotechnology Research in Medicine).
The SARS-CoV-2 infection and its RNA polymerase
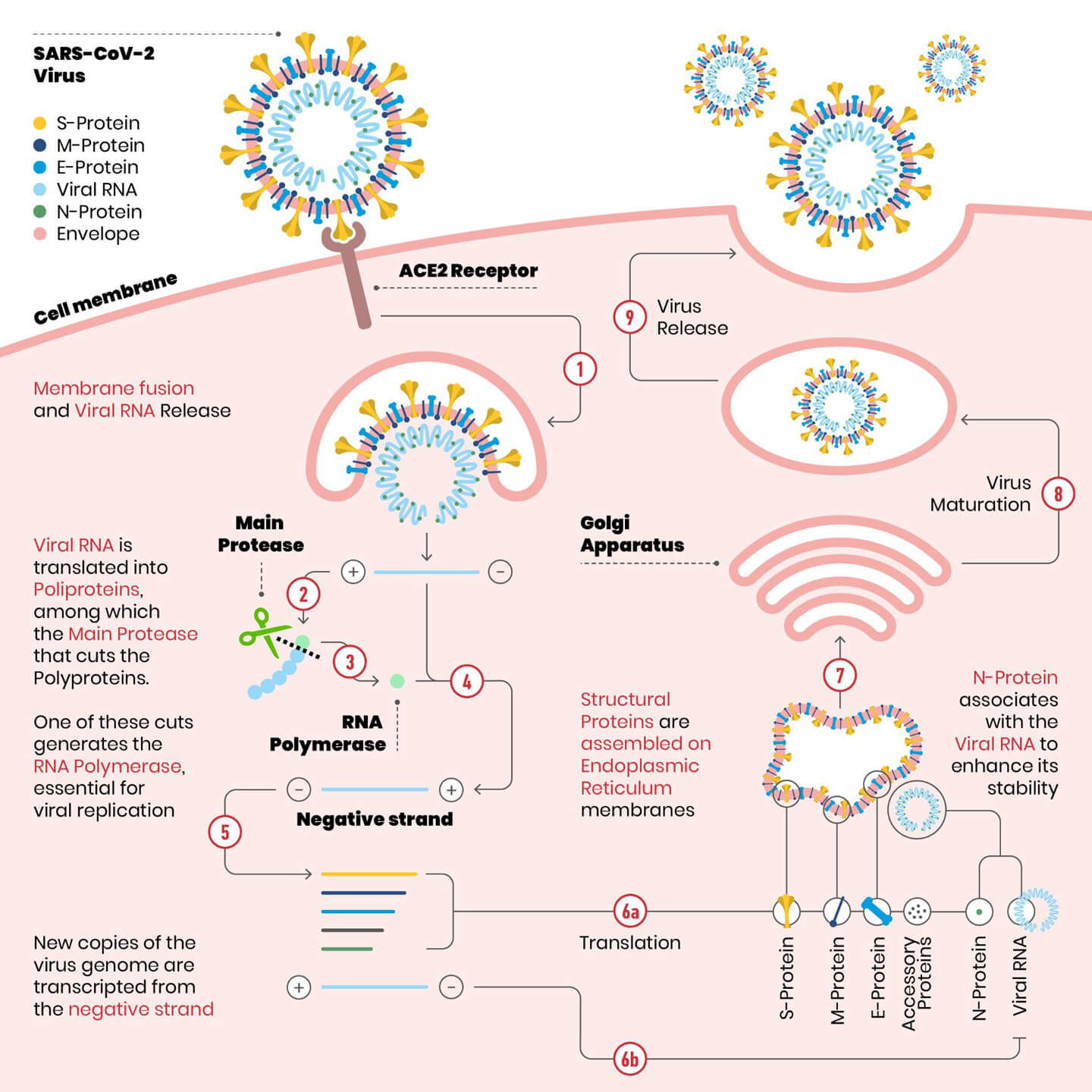
As we have previously seen, when a viral infection occurs, the virus enters the host cell, initially docking through a viral receptor (in the case of SARS-CoV-2, this is the Spike protein) that hooks to a protein of the target cell (in the case of SARS-CoV-2, this is the ACE2 enzyme). Once inside the aqueous part of the cell, called cytosol, the virus “unpacks” itself releasing its content: a mixture of proteins and its genetic material. While genetic information, i.e. what is necessary to build cell components, is stored in DNA molecules in humans, Coronavirus uses a single RNA molecule. Humans also have RNA in their cells, but we use it for other purposes, primarily to build proteins.
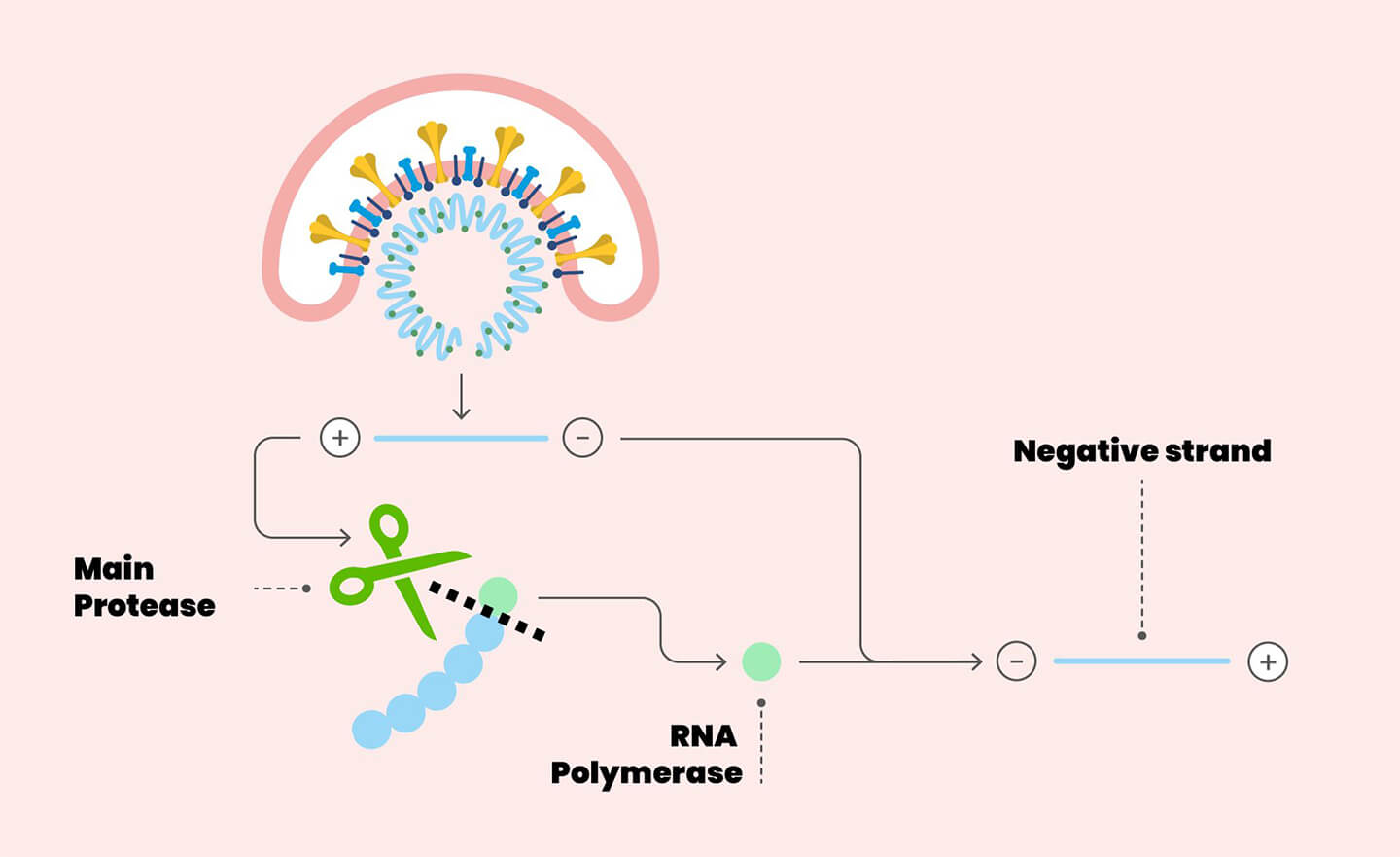
Once the coronavirus has infected a cell, its purpose is to ensure that new copies of the virus are formed, in order to have offspring capable of perpetuating its species. Therefore, all that was present in the virus that infected the cell must be “copied” and assembled to form new virions, including new copies of the genetic material (=RNA) of the virus itself. The essential machinery for synthesizing new RNA is found, in different forms, in all organisms and is called RNA polymerase, literally: “enzyme that forms long chains of RNA”. The SARS-CoV-2 RNA polymerase is called RdRP (RNA-dependent RNA polymerase), essentially copying the virus’ genetic material.
A “sloppy photocopier”
Despite carrying out a sophisticated and crucial process, the machinery (or the operator) is somewhat “careless”: in fact, every time it makes a copy of the original genetic material, it makes errors, and the copy has differences compared to the original. These differences are the so-called mutations, that affect the structure of the virus proteins and their function. In recent days we have heard about the G614 (or better, D614G) variant of the virus: what happened is that the RNA polymerase got “distracted” while making a copy of the viral genome, and it has changed one base (a “brick”) of RNA. This caused a mutation of the Spike proteinin position 614, which however is not a site of direct contact with the ACE2 enzyme, so its effect on the structure and function will have to be assessed experimentally to understand its consequences. In any case, these mutations have consequences on the virus, meaning they can be neutral, advantageous or disadvantageous. A mutated virus will begin to circulate if this mutation gives it an advantage for its life cycle: this may mean being more efficient at infecting human cells, for example, but causing less damage to the host organism, as a highly lethal virus it is destined to disappear for lack of guests.
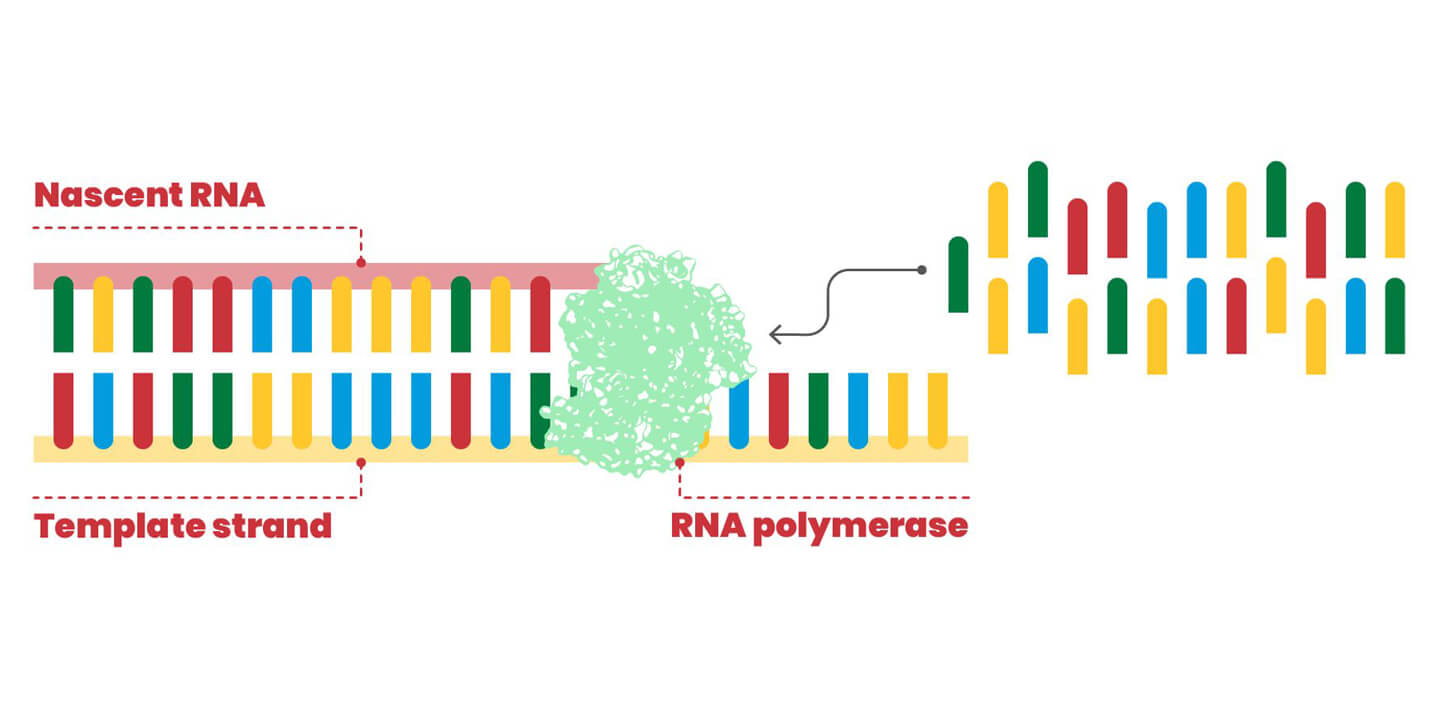
The RdRP enzyme synthesizes a copy of the genetic material of the virus, an RNA molecule made up of four different nitrogen bases. The enzyme takes the RNA strand to be copied and the individual molecular units (nucleotides) complementary to it appear one at a time according to the A:U and G:C pairing (in the figure, the rectangles with different colors pair yellow with blue and red with green). Sometimes RdRP mismatches these bricks, and this leads to a mutation in the viral "offspring". If the mutation is beneficial for survival, it will be maintained.
The antiviral Remdesivir
Let’s go back to the primary function of the RNA polymerase, i.e. to copy the genetic material of SARS-CoV-2. This is a crucial step to form a new virus, and therefore if we could block the action of RdRP in the infected cells... you are correct, the virus would stop replicating. Molecules capable of inhibiting RNA polymerase are excellent antivirals! The Remdesivir molecule, developed to inhibit the Ebola virus RNA polymerase but also capable of blocking the polymerases of other viruses, is an excellent example of antiviral targeting RdRP. Similarly to what has been seen for the main protease Mpro of SARS-CoV-2, structural biology can tell us how Remdesivir can jam the machinery, and most importantly it suggests how to improve the molecule to ensure that a highly specific antiviral for coronavirus can be obtained. Researchers from the Chinese Academy of Sciences have determined, using the technique of cryoelectron microscopy, the structure of RdRP, photographing it in the midst of its replicative activity.
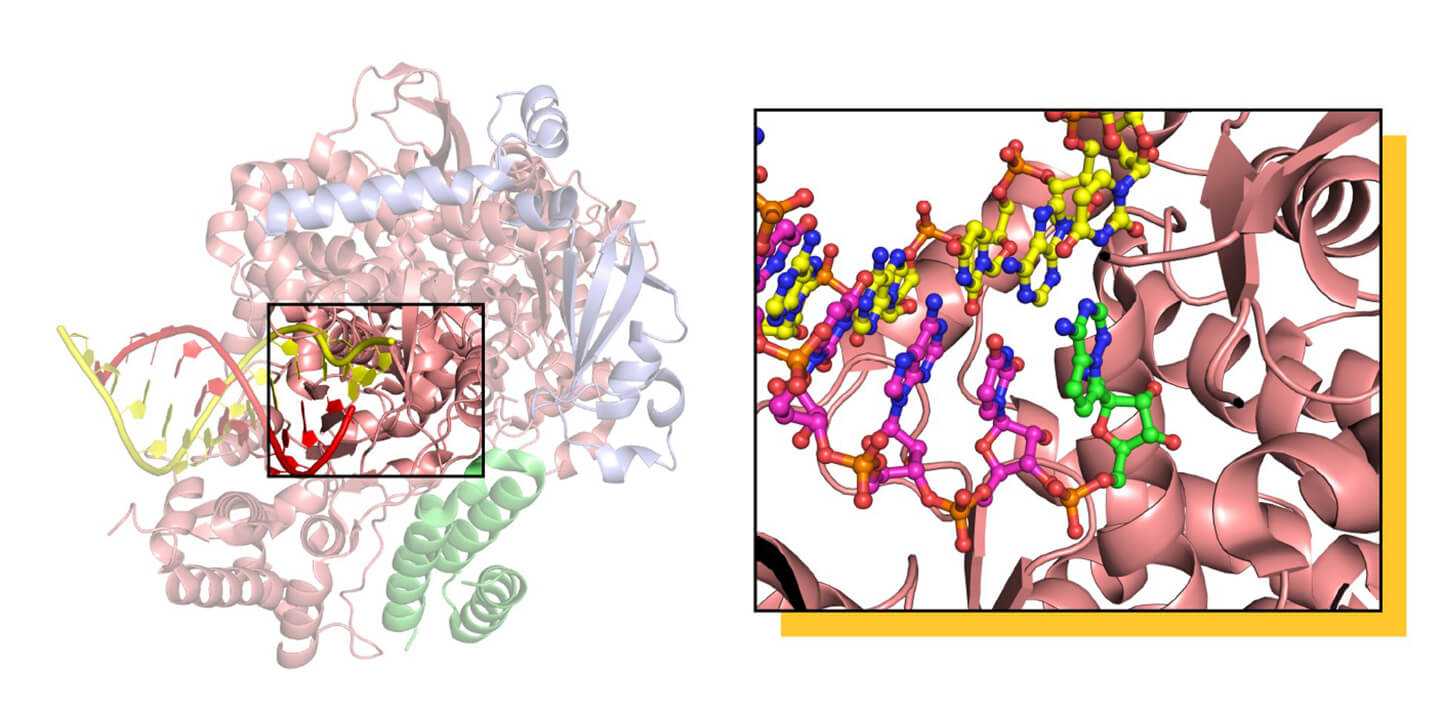
The structure of RdRP from SARS-CoV-2, composed of three protein subunits to form an active heterotrimer, captured by cryo-EM in the RNA replication process, visible on the left side. The heart of the RdRP machine is highlighted on the right, where the active form of the drug Remdesivir is displayed in green, incorporated in the copy of the genetic material of the virus. The addition of the drug to the copy of the RNA prevents the complete copying of the genetic material, and the fine details of the mechanism are still under debate. Courtesy of Dr. Degano.
This structure has shown that Remdesivir acts in a very particular way, not so much by jamming RdRP, but by inserting itself into the copy of the RNA that is created and acting there as a “stop” signal, preventing the work of the RNA polymerase from continuing. Even more interestingly, the structure has shown that the point where Remdesivir binds to RdRP before being incorporated into the RNA has very specific characteristics that can be used by chemists to synthesize an even more efficient antiviral molecule. Two additional structures determined by other groups in different conditions offered other information on the mechanism of action of the anti-viral and on how to develop better ones. At present, there is already a compound called EIDD-2801 which is up to 10 times more potent than Remdesivir in blocking SARS-CoV-2 replication in cultured cell. There is still a long way to go for a drug to be available, but thanks to structural biology, we are making great strides.