Electrical cryomicroscopy: small electrons to visualize large molecules
Publication date: 07-07-2020
Updated on: 16-05-2025
Topic: Covid-19
Estimated reading time: 1 min
Article Author
Eufemia PutortiMedical Editor
Massimo Degano
Editor and Translator
Viktoryia LuhakovaThe ongoing COVID-19 pandemic has forced the scientific community to devote itself to the research and development of various strategies to fight SARS-CoV-2. The only way to identify new antivirals is through the knowledge of the three-dimensional structure, the shape, of the virus proteins that are responsible for cell infection and virus replication. Once the structure of proteins is known, it is possible for chemists to synthesize compounds that block its function, and consequently the virus’s life cycle. We discussed the matter with Dr. Massimo Degano, Group leader of the Biocrystallography Unit at the San Raffaele Reasearch Hospital and lecturer at the Vita-Salute San Raffaele University.
The cryo-electron microscopy (cryo-EM) allows observing in atomic detail the structure of molecules of infinitesimal dimensions: what does it consist of?
Electron microscopy is substantially similar to optical microscopy, which works by irradiating an object with visible light and re-focusing the rays deflected by the object through lenses on the eye retina or on a screen. By using appropriate lenses, we can enlarge the image to appreciate the finer details. Here the term “resolution” comes into play, i.e. the ability to see two objects as distinct at a minimum distance. The human eye has a resolution of approximately 0.1 millimeters. With optical microscopes that exploit visible light it is possible to reach a resolution of 0.2 micrometers, that is 500 times greater. Such resolution allows observing individual cells, but not smaller objects such as viruses, proteins and other biological molecules.
To see even smaller objects, you need to use something other than visible light: electrons. Electrons, subatomic particles and therefore much smaller even than the molecules we want to visualize, are deflected from their path by the atoms of the molecules, exactly as light is deflected by the edges of an object. With very specific lenses, made not of solid materials but made up of electric fields, we can focus the electrons and obtain the image of the molecules.
Described as such, the technique may seem trivial. But there are many technical issues that have limited their use for years:
- firstly, the molecules in the sample move, therefore the image is blurred as in a photograph, greatly limiting its resolution;
- secondly, electrons are particles with high energy and damage the molecules that they “light up”. All this has been overcome by the introduction of cryo-electron microscopy, in which the sample is cooled up to -173.15 °C: at this temperature water becomes glass-like, the molecules are immobilized and the damage caused by electrons is also reduced (discovery awarded with a Nobel Prize).
A schematic representation of the cryo-EM experiment is shown below.
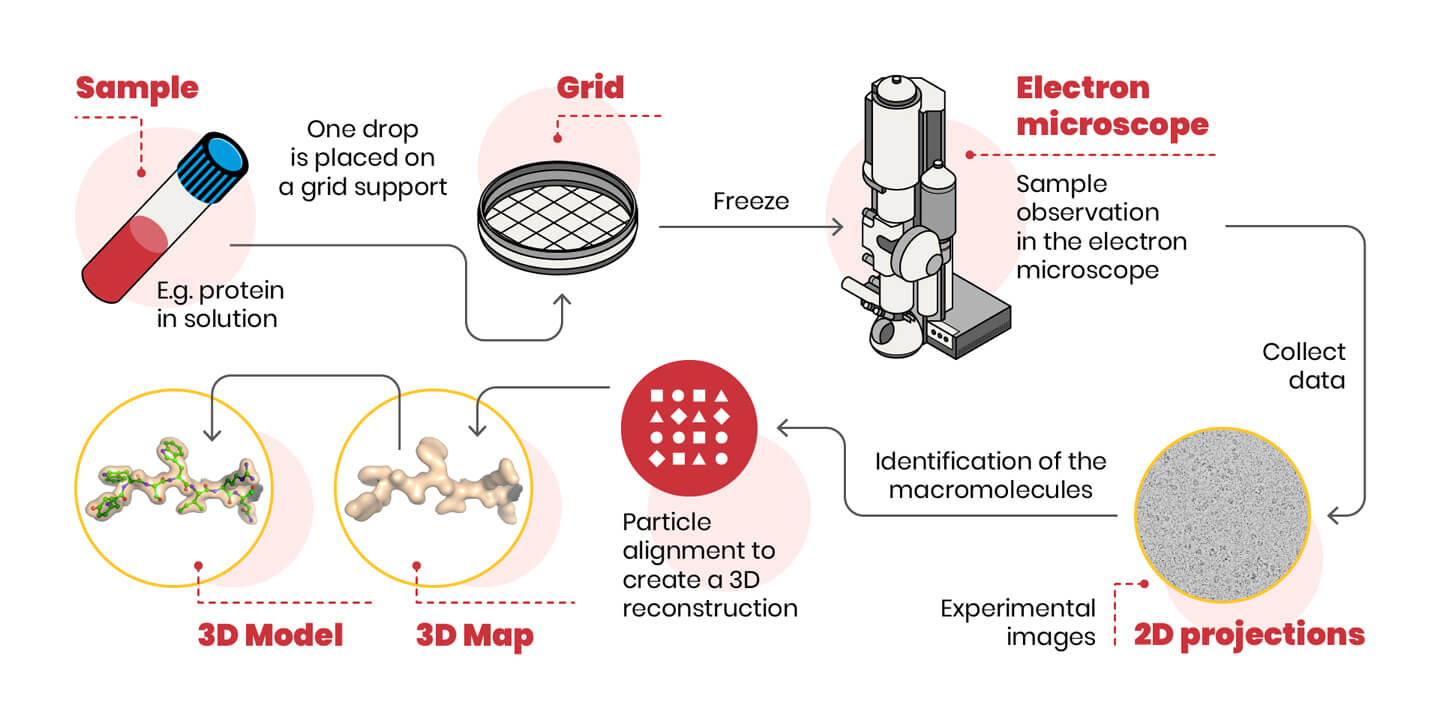
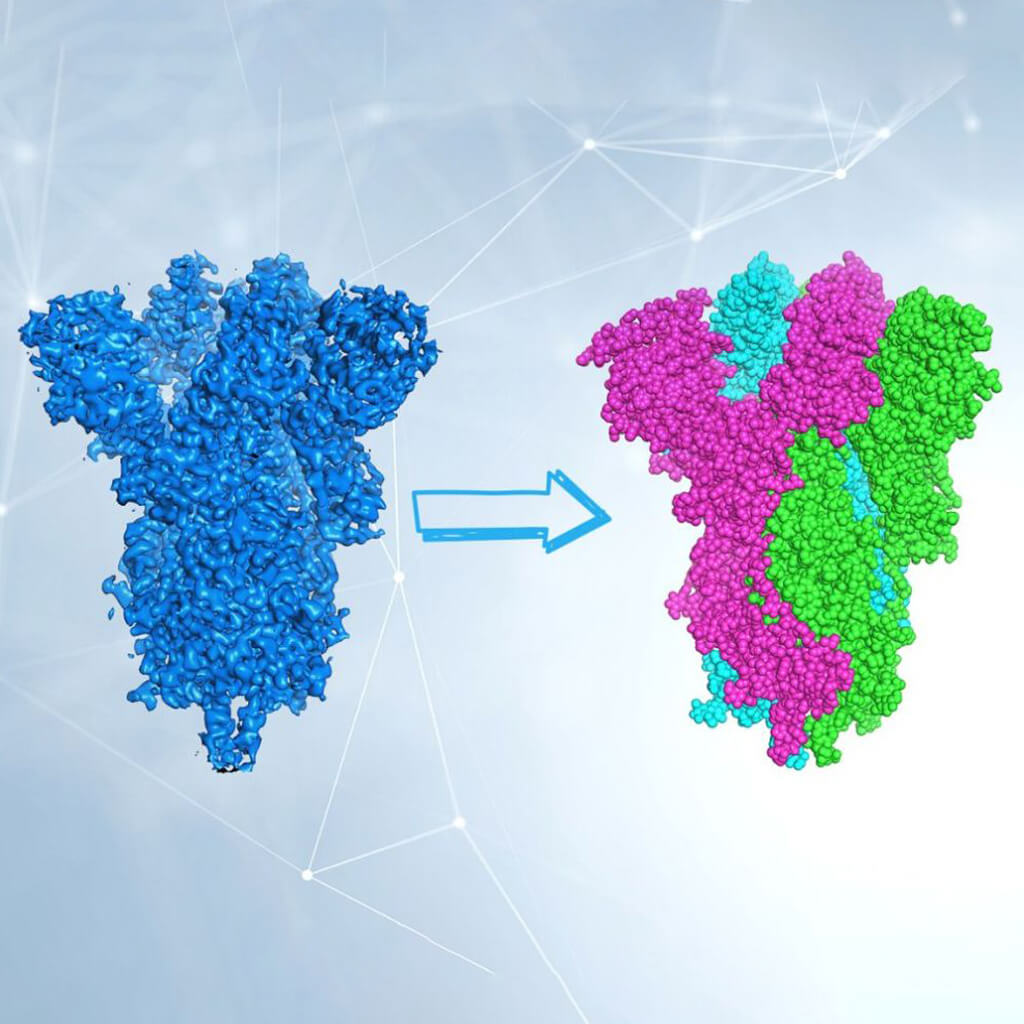
One last difficulty remains: we are able to appreciate the three-dimensional image of an object also because we can look at it from multiple angles. In cryo-EM the object (the molecule) is photographed oriented in many different ways. Therefore, 2D images of randomly oriented molecules in space are used to reconstruct, through sophisticated computer algorithms, the 3D shape of the molecule. Now it’s up to biochemists to interpret electron density and build the protein model.
Cryo-EM is a cutting-edge and extremely powerful technique, and in the last few years has allowed us to determine the structure of molecules that have long represented the holy grail for many researchers. The structure of the SARS-CoV-2 Spike protein has been determined a couple of months after the sequencing of the viral genome, and allowed a fine understanding of its interactions with the ACE2 cell receptor. Like any technique, however, it has limitations: the molecules being studied, for example, must be quite large. Another limit, not negligible, is the cost of the microscope itself and its maintenance.